Accelerated Aging Diseases
Running research topics:
Progeroid disorders
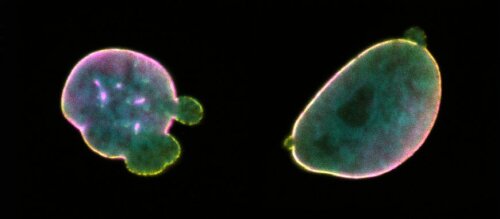
Laminopathies are genetic diseases that target specific tissues such as adipose, muscle or nerves, but can also act systemically to induce progeria or metabolic syndromes. Despite the bewildering diversity of disease manifestations, all these pathologies share a common cellular feature: defects of the nuclear lamina, a filamentous network that delineates the cell nucleus. How these defects contribute to disease development is, however, not yet fully understood. With an eye on novel diagnostic and therapeutic opportunities, we aim to enhance the fundamental understanding of laminopathies from a cell-centered perspective. Currently, we are establishing isogenic cell lines that express mutant lamins using innovative genome editing technology. Alike patient cells, model cells are fingerprinted at a molecular level by means of next-generation sequencing and spatial proteomics approaches. Interesting hits from these experiments are monitored in cellula by high-content, live cell imaging and label-free microscopy.
Neurodegeneration
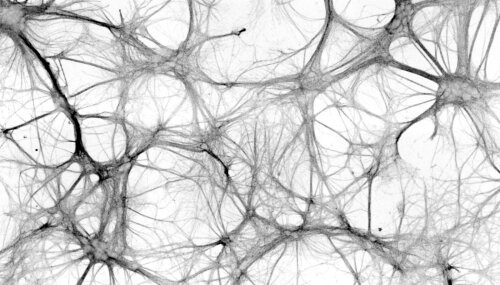
Brain function relies on an intricate network of neuronal connections. These connections are far from static. Indeed, neurons remodel their morphology dramatically under impulse of various external cues and pathological conditions. Among the neuronal structures that demonstrate morphological plasticity are neurites, synapses, dendritic spines and even nuclei. Their remodelling is directly connected with functional changes such as inter-cellular communication and the adjoined calcium-bursting behaviour. We use quantitative high-content microscopy and image informatics to interrogate neuroplasticity in neuronal networks derived from primary hippocampal cultures or induced pluripotent stem cells. We focus on the underlying cell biological mechanisms and also explore potential therapeutic strategies for modulating neuroplasticity.
Neuroinflammation
Inflammation plays a key role in the etiopathogenesis of neurodegenerative diseases through altered activation states of microglia and macrophages (M1, M2, …). Progranulin involved in frontotemporal lobar degeneration is a pluripotent growth factor. Utlizing progranulin conditional knockout mice and neuronal and microglial cell cultures, we aim to understand how progranulin is involved in tissue homeostasis and preserves M2 phagocytic functions
Relevant publications
Janssens,J., Wils,H., Kleinberger,G., Joris,G., Cuijt,I., Ceuterick-de Groote,C., Van Broeckhoven,C., & Kumar-Singh,S.: Overexpression of ALS-associated p.M337V human TDP-43 in mice worsens disease features compared to wild-type human TDP-43 mice. Molecular Neurobiology 2013 Aug;48(1):22-35. (2013)
Wils,H., Kleinberger,G., Pereson,S., Janssens,J., Kleinberger,G., Van Dam,D., Joris,G., Cuijt,I., Smits,V., De Deyn,P., Van Broeckhoven,C., & Kumar-Singh,S.: Increased TDP-43 phosphorylation and liver pathology in progranulin knockout mice. Journal of Pathology 228(1):67-76 (2012)
Kumar-Singh. S, Progranulin and TDP-43: mechanistic links and future directions. J. Mol. Neurosci. Special issue on frontotemporal dementia J. Mol. Neurosci. Nov;45(3):561-73 (2011)
Kleinberger,G., Wils,H., Ponsaerts,P., Joris,G, Timmermans,J.P., Van Broeckhoven,C., Kumar-Singh,S. Increased caspase activation and decreased TDP-43 solubility in progranulin knockout cortical cultures. J Neurochem. 115(3):735-47 (2010) (I.F.:4.206)
Wils,H., Kleinberger,G., Janssens,J., Pereson,S., Joris,G., Cuijt,I., Smits,V., Ceuterick-de,G.C., Van Broeckhoven,C., & Kumar-Singh,S.: TDP-43 transgenic mice develop spastic paralysis and neuronal inclusions characteristic of ALS and frontotemporal lobar degeneration. Proceedings of the National Academy of Sciences of the United States of America 23(107), 3858-3863 (2010) News: Going Wild About the Latest TDP-43 Mouse Model on Alzheimer Forum or ALS forum)