-
 Mother and child participating in the trial
Mother and child participating in the trial
Boende, DRC - The PregInPoxVac project is entering a new phase: the trial has successfully completed enrolment of all 363 pregnant participants at the end of January 2026, while in October 2025 the infant cohort reached 344 participants, meaning both key groups have now reached full enrolment.
Achieving full enrolment reflects the outstanding collaboration among all consortium partners involved in this multi-stakeholder project. The dedication shown by the entire project team ensured that the study remains firmly on track.
Prof. Hypolite Muhindo Mavoko (UNIKIN): "Recruiting pregnant women and infants in such a remote area was challenging, but we made it. We had to address concerns related to vaccine rumours, fears around blood sampling, and the influence of religious and family beliefs, in addition to logistics complexities. Through close collaboration with community leaders, local mobilisers, and health services, and by clearly communicating the purpose of the study, we sustained trust (built from our previous trials) and supported informed participation among families."
After extensive informed consent procedures and thorough screening, pregnant women were invited to participate in the study during their second or third trimester. They were then randomly assigned to receive the vaccine either during pregnancy or shortly after giving birth. Once the mothers deliver, samples such as maternal blood, cord blood and breastmilk are collected. Until one year after delivery, the mother and baby will return for several planned visits, allowing the team to closely monitor their health and help us better understand how immunity against mpox develops following vaccination, as well as how mothers pass antibodies on to their newborns.
The cohort studying infants (aged 4 to 24 months) is divided into two groups receiving different vaccine doses. This study focuses on understanding how young children could respond to vaccination and mount an immune response after different dosing strategies.
Prof. Jean-Pierre van geertruyden (UAntwerp): “Setting up this trial required a tremendous effort to establish all necessary enablers - sensitisation activities, standard operating procedures, electronic case report forms, ancillary care procedures - that make such complex research possible. Yet, a long journey still lies ahead, as the women and infants will be followed until one year after birth to assess vaccine-induced immune response in these vulnerable groups – populations that are too often neglected in clinical research. Reaching full enrolment is a key milestone, and I am both proud and deeply grateful to our teams in DRC, Kenya, Italy and Belgium, whose close collaboration across continents is making this ambitious study a reality.”
With both trial cohorts complete, the PregInPoxVac project enters a critical phase of follow-up and analysis; bringing the field closer to understanding how maternal and early-life immunisation may contribute to mpox prevention.
The Consortium expresses gratitude to CEPI and EDCTP for funding this project.
-
 Group photo
Group photo -
 The PregInPoxVac site
The PregInPoxVac site -
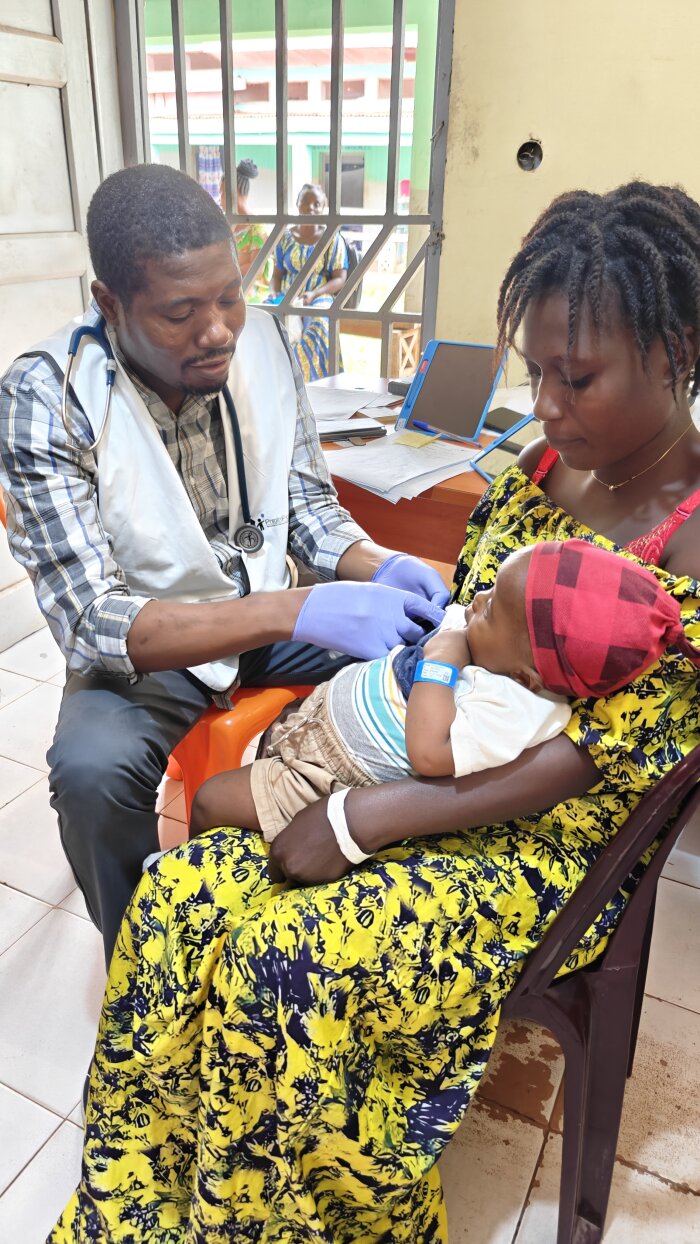 Mother and child participating in the trial
Mother and child participating in the trial -
 Mother and child participating in the trial
Mother and child participating in the trial -
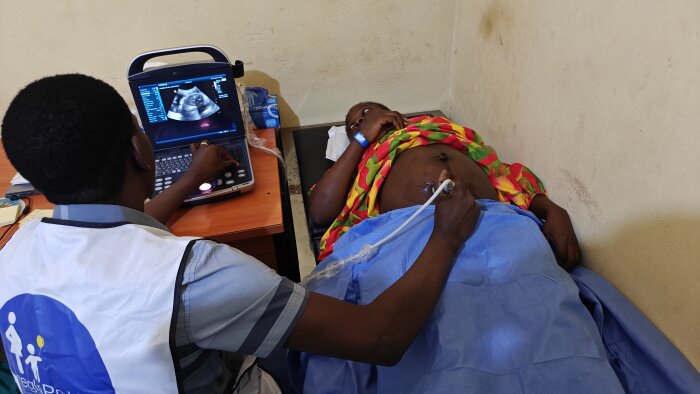 A participant during a doctor's appointment
A participant during a doctor's appointment -
 PregInPoxVac
PregInPoxVac
