Next-generation Microscopy
Running projects on microscopy technology:
Deep coverage microscopy
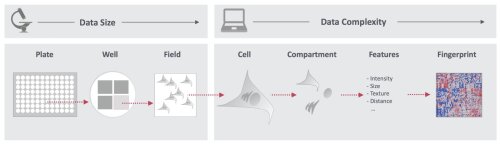
No longer solely descriptive, microscopy has turned into a mature, quantitative technology, amenable to upscaling. To meet a growing need for better mechanistic insights and translatability of drug screens, we aim at establishing robust microscopic imaging pipelines that enable cellular phenotyping with high fidelity and statistical power. The emphasis of the developments lies in acquiring large data sets (high-throughput) that are rich in information (high-content). We deploy these deep coverage imaging strategies in studies of neurodegeneration, laminopathies and exposome screens.
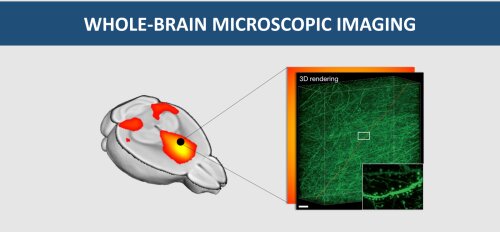
In toto imaging
Numerous neurodegenerative disorders are typified by an accumulation of misfolded proteins. Where those aggregates form and how they spread is not yet clear, nor is it known how the diseases affect neuronal function and wiring. Enhanced insight in the spatiotemporal correlation of functional and structural aspects of brain defects is urgently needed to enable early prognosis. However, current methods to study the mammalian brain involve sectioning and reconstruction, which are prone to artefacts and are not always compatible with molecular phenotyping. That is why we are working towards in toto imaging approaches that allow registration of intact brains with molecular resolution. Implementation of chemical clearing methods (e.g. CLARITY) will extend the penetration depth for light microscopy by reducing scattering, while preserving epitope integrity. Complementary to that, we are building a digitally scanned light sheet microscope, allowing large object imaging with unprecedented speed and minimal photobleaching. The holistic approach of intact brain imaging will not only be instrumental for bridging the gap in resolution between the cellular and the tissue level but will also find a ready market in preclinical drug development, for instance for monitoring the blood-brain-barrier penetration of therapeutic compounds