Algology (Pain Research)
Research topics
- Trigeminal neuropathic pain (IoN-CCI model)
- Endothelin-1 (ET-1) in cancer pain
- Electrocorticographic (EcoG) recordings in freely moving awake rats
Trigeminal neuropathic pain
Neuropathic pain syndromes result from damage or dysfunction of the somatosensory nervous system. They are very resistant to common pain-killers and patients often suffer from terrible discomfort. Trigeminal neuralgia (“tic douloureux”) is an extreme form of facial neuropathic pain.
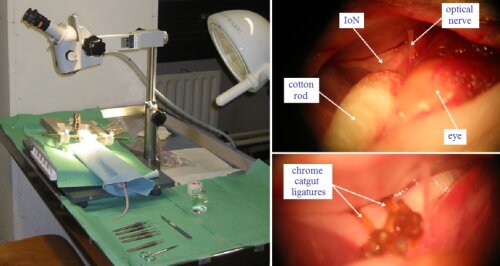
A rat model was developed where the Infraorbital nerve (IoN), which constitutes almost the entire maxillary branch of the trigeminal nerve, is loosely ligated. Following IoN surgery, rats display increased face grooming behavior directed to the territory of the injured IoN indicative of spontaneous pain.
A distinction is made between between
- Face grooming during body grooming, which is part of normal grooming behavior and which remains unaltered by IoN surgery, and
- Isolated face grooming, which is specifically related to IoN-CCI induced spontaneous pain.
After an initial period of hyporesponsiveness, rats also become hyperresponsive to mechanical (von Frey hair) stimulation of the IoN territory. A clear distinction can be made between
- Mild responses to mechanical stimulation of the contralateral IoN territory, and
- Strong responses to mechanical stimulation of the ipsilateral IoN territory.
A video summary of the IoN-CCI model can be found at http://www.jove.com/video/53167

Endothelin-1 (ET-1)
In addition to its role in tumourigenesis and angiogenesis, evidence is accumulating that ET-1 may be of critical importance in the occurrence of cancer pain.
We found that ET-1 produces endothelin receptor-dependent pain behaviour when applied to the rat sciatic nerve, i.e. unilateral, hind paw flinching lasting 60 min. Pre-emptive systemic morphine completely blocked this effect in a naloxone-reversible manner. In a next step, impulse activity of single, physiologically characterized sensory C-, Ad-, and Ab-fibers was recorded from the sciatic nerve in rats after subcutaneous injections of ET-1. All nociceptive C-fibers were excited by ET-1 in a dose-dependent manner. All nociceptive Ad-fibers also responded to ET-1. In contrast, most Ab-fibers did not respond to ET-1. These data demonstrate that subcutaneous administration of ET-1 produces selective excitation of nociceptive fibers.
Recently, our lab has been focusing on the effects of application of ET-1 to the infraorbital nerve (IoN) of the rat and of prolonged exposure to ET-1. Despite the multitude of recent studies, still very little is known about its neurophysiological effects during chronic exposure. Indeed, until now all animal and human studies have exclusively studied the effects of acute (in most cases, single) administration of ET-1. Cancer patients, however, are continuously exposed to high(er) levels of ET-1, either systemically or locally in the surroundings of the tumour. Therefore, a critical next step in the investigation of the pathogenesis of ET-1 induced cancer pain syndromes, should be targeted towards the identification of neurophysiological changes occurring after prolonged exposure to Endothelin-1.
ECoG recordings
Video-ECoG, or the simultaneous recording of behaviour and electrocorticography in awake animals, has become a valuable tool in neurophysiological and neuropharmacological studies. In our lab, we have begun to implement the video-ECoG methodology in the IoN-CCI model in order to correlate behavioral (grooming) data with ECoG data in normal and IoN-CCI rats. Two trigeminal neural structures are interesting targets for EEG recording: the ventral posterior medial (VPM) thalamic nuclei and the somatosensory cortex. In both areas, neural responsiveness was found to be significantly altered following IoN-CCI.